The last 14 months have been a wild ride. The desire to provide employees, patients, customers and/or family members with a virus-free environment fueled an explosion of products with questionable efficacy against SARS-CoV-2, the virus that causes COVID-19.
The CDC recently released guidance clarifying the role of contaminated surfaces in the spread of SARS-CoV-2. The CDC’s position is that the primary method of transmission of SARS-CoV-2 is through direct contact, droplet or airborne transmission; and that indirect transmission plays a relatively minor role. However, the CDC has determined that there are situations where risk for surface transmission still exists for up to 3 days after the surface was contaminated. The CDC states, “In situations when there has been a suspected or confirmed case of COVID-19 indoors within the last 24 hours, the presence of infectious virus on surfaces is more likely and therefore high-touch surfaces should be disinfected.”
The CDC’s guidance further states that “…the surface must be treated with a disinfectant product registered with the Environmental Protection Agency’s (EPA’s) List N or technology that has been shown to be effective against the virus.” Products that do not meet this standard may not be providing adequate disinfection.
Fogging/misting/electrostatic sprayers have become popular for disinfection of large areas, primarily because of their low cost. Regarding these products, the CDC guidance states, “Some types of disinfection applications, particularly those including fogging or misting, are neither safe nor effective for inactivating the virus unless properly used.” Additionally, the EPA recommends against using them unless the disinfectant being used has specifically been approved on List N for use in a fogging system.
So where does that leave us in regard to fast and effective methods for room disinfection? The EPA does not include UV disinfection devices on List N, so these systems must be tested against SARS-CoV-2 in the laboratory environment to prove efficacy for surface disinfection. Xenex’s LightStrike Robot and Deactivate portable LED disinfection unit were both tested against SARS-CoV-2 at Texas Biomedical Research Institute, a world-renowned research facility, and shown to deactivate the virus in 2 minutes at 1 meter for LightStrike and 30 seconds at 1 meter for Deactivate, respectively.
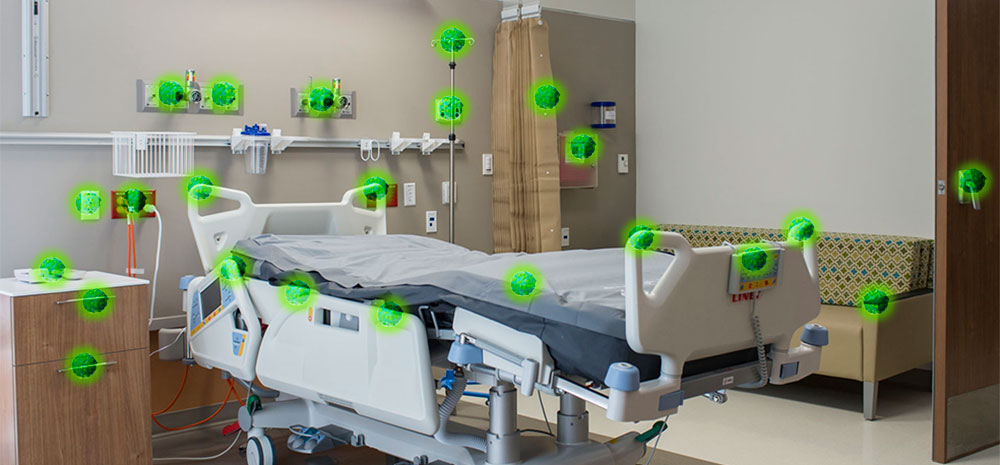
One of the settings where environmental disinfection will continue to be an important focus is healthcare. Even before the COVID-19 pandemic, contaminated surfaces were known to be a problem in hospitals. MRSA, C. difficile, and a variety of drug-resistant gram-negative bacteria can be passed from patient to patient through high-touch surfaces like bedrails, tray tables, nurse call buttons and doorknobs that are not adequately disinfected using traditional cleaning methods.
LightStrike robots have been deployed in over 800 facilities worldwide to provide fast and effective disinfection. The need to combat the presence and spread of pathogens that can lead to infections still exists and antibiotic resistance is a real threat. Assuring a disinfected environment will remain a priority for healthcare facilities well into the future.