On any given day, we can turn on the TV, radio, or peruse social media outlets and hear about new research that has concluded something powerful, helpful, or frightening. But what do you really know about that study? Where was it conducted? Did something influence the results? Do you know where it was published so that you can read it yourself?
Weeding through the “evidence” that exists about disinfection science is equally challenging. If we don’t know the right questions to ask about a “study” on new technology, we can’t be certain the claims are legitimate or that the study validates the use of the technology in healthcare.
Fortunately, a vetting system already exists within the academic world. Think of it as a staircase that must be climbed to take data and establish it as scientific fact.
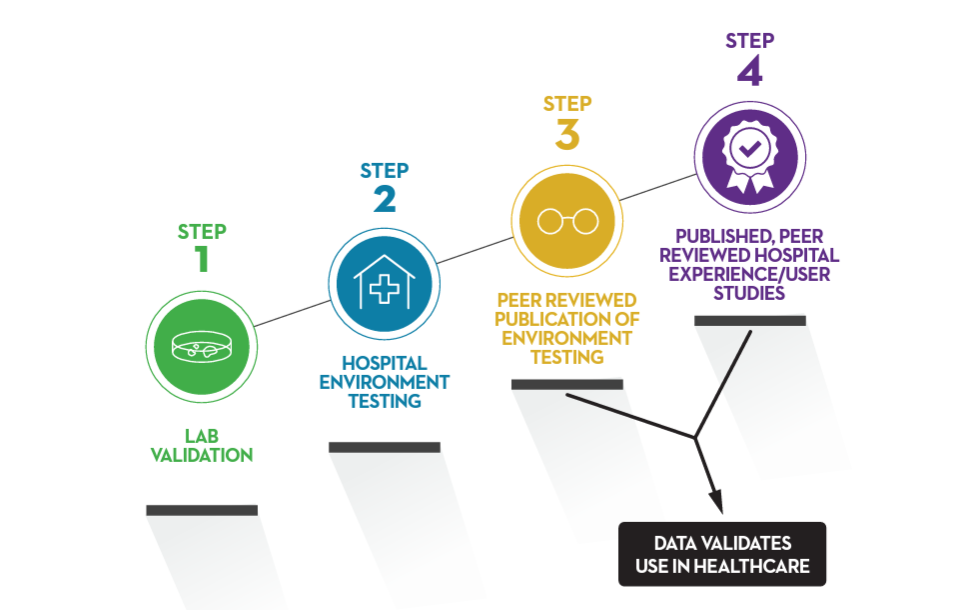
So, why are these steps important when evaluating technologies? Because it shows the level of evidence a product has achieved. Sometimes you’ll find products using research from a totally different product but applying the results to theirs. This is risky and can be speculative since the claim they are making about their product came from a study using a different product.
Let’s discuss what each of these steps actually mean.
Step 1: Lab Validation
This is the initial step in proving the theory that the technology can actually achieve what it is intended to do.
This type of research is conducted at an independent, 3rd party, Good Laboratory Practice (GLP) certified microbiology laboratory. The lab will use the technology in the manner it is intended to be used by the manufacturer to collect data.
To pass, the results must be consistent across a large number of tests and are typically presented as a % reduction in the pathogen or as a “log reduction.”

Step 2: Hospital Environment Testing
If your technology had success in the lab, now it’s time to see how it performs in an environment that is similar to the one it will be used in. It’s still a lab, but now it looks like a typical hospital room.
This step incorporates the challenges of the real world environment in a room with a known pathogen to evaluate how the technology performs on a true pathogen load. Clear sampling protocols are used to ensure methods are consistent for data collection, the number of samples to be collected are identified, and the steps that will be taken to analyze this process are clearly defined.
To pass, the results show statistical significance (a p-value) that tells us we can correlate the results to the disinfection technology and that they are not due to chance.
Step 3: Peer Reviewed Publication of Environmental Testing
When environmental lab testing yields positive results (clear statistical significance), it’s time to submit for publication.
In this step, a group of highly qualified, impartial scientists reviews the new findings before they are considered truly valid. In most cases, this will be with a known industry journal that has a team of specialists who examine submitted studies and complete the “peer review” process.
Making it through the peer review process is not an easy task. Your research must be thorough and you need to show that your process is repeatable. There are strict ethical guidelines and your study cannot have any expression of opinions – just the facts.
Step 4: Published Peer Reviewed Hospital Experience/User Studies
This is the Gold Standard of Disinfection Science. This step is achieved when the disinfection technology shows a measurable, statistically significant pathogen reduction in the environment it is intended to be used. This type of research is typically conducted by a facility that has integrated the technology.
The results from a study are examined through the same lens as Step 3, as well as:
- A thorough review of pathogens of concern and a correlation with historical pathogen reduction
- Identification of any other interventions that may be responsible for the improved outcomes, like hand-washing initiatives
While many UV disinfection technologies have achieved steps 1 and 2, and a few have achieved publication, LightStrike remains the only UV disinfection system with over 45 studies published in peer-reviewed journals.
Lastly, we suggest this should be part of your UV evaluation process.
Evaluating all of the published research on the product you’re evaluating is a great way to uncover questions to ask and validate important factors like:
- Which of the above evidence steps has the product achieved?
- Was the technology you’re evaluating the same technology used in the study?
- What was the disinfection cycle time and distance and is that repeated in more than one study?
- Is there a different cycle time for different pathogens like vegetative bacteria vs spores?
- What was the sample size for the study (more are better)?
- Was the study conducted in the environment you will be using the technology in (patient rooms, operating rooms, emergency department, etc)?


















